Naming CHO Cells for Bio-manufacturing
Genome Plasticity and Variant Phenotypes of Cell Populations in Bioreactors Question the Relevance of Old Names
Author's note
“What cells do you work with?”
When scientists involved in biomanufacturing get to know each other, this is one of the first questions they ask. Each secretly hopes the other doesn’t have “better cells”.
Cell names such as CHO–K1, CHO DG 44, CHO DUX, etc. have been in use for more than 40 years. In this review, Maria and Florian try to summarize the complexities of genomes of cultivated CHO cells. Pointedly, they question the relevance of “old names” which are used in the context of today’s manufacturing activities.
To do so, the authors revisit the history of these cells. They also discuss several insightful publications from the last 30 years and reveal unequivocally that CHO cells in culture undergo rapid evolution by restructuring their genomes. This means that the cells used today are drastically different from the cells established and named 40 to 60 years ago! A new naming convention is suggested to better represent cell populations in well-established banks and highlight phenotypic features that are useful to the industry.
The paper was published in Biotechnology Journal in July 2021, and featured on the title page with an image that illustrates some of the complex cytogenetic events that occur frequently in CHO cells, often without the user’s knowing.
Paper abstract
Chinese Hamster Ovary (CHO) cells are the workhorse for production of modern biopharmaceuticals. They are however immortalized cells with a high propensity for genetic change. Judging from published culture records, CHO cell populations have undergone hundreds of population doublings since their origin in the late 1950s. Different cell populations were established and named from 1 to 3 decades after their generation, such as CHO-Pro–, CHO-K1, CHO-DG44, CHO-S, CHO-DUK, CHO-DXB-11 to indicate origin and certain phenotypic features. These names are commonly used in scientific publications still today. This article discusses the relevance of such names. We argue that they provide a false sense of identity. To substantiate this, we provide the long (and poorly recorded) history of CHO cells as well as their highly complex genetics. Finally, we suggest an alternative naming system for CHO cells which provides more relevant information. While the implementation of a new naming convention will require substantial discussions among members of the relevant community, it should improve interpretation and comparability between laboratories. This, in turn will help scientific communities and industrial users to attain and further the full potential of CHO cells.
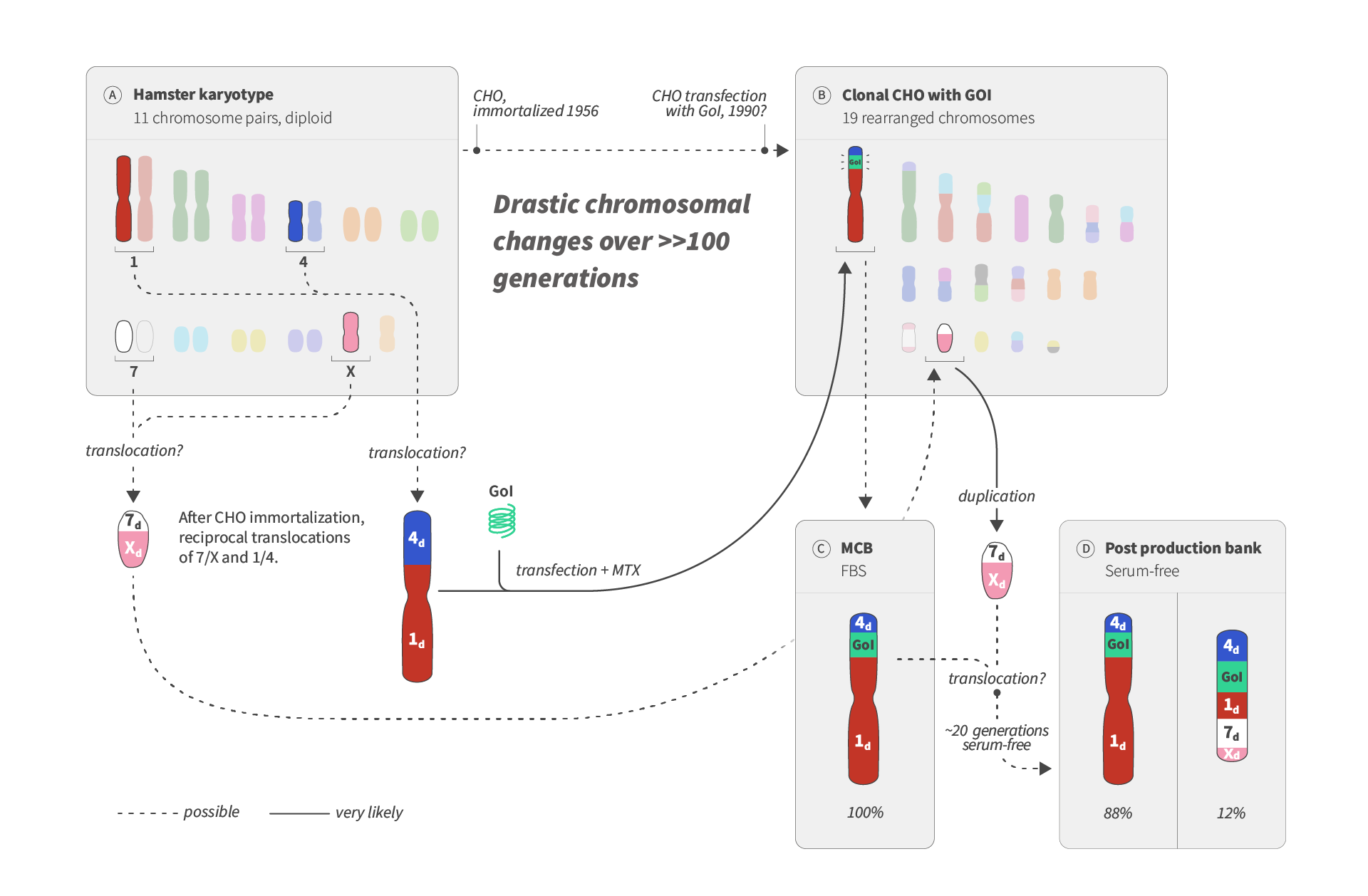
Visual abstract of karyotype variability in CHO cells
CHO cells have a long history. They were derived from Hamster cells containing 11 pairs of chromosomes and have since been used to produce biopharmaceuticals — for fundamental research and for production in large-scale bioreactors. The image provides a simplified example of the complex genetics of CHO cells (first published by Roullier et al., 2015).
In cultures, chromosomes will tend to rearrange structurally, and this results in drastic genome changes over time. Why? Darwinian evolution seems the most likely explanation.
When cells are transfected, the Gene of Interest (GoI) DNA integrates into the chromosomes of the cells. In the example, the eventually established Master Cell Bank (MCB) showed 100% of analyzed cells contain a large chromosome where the GoI is framed by a fragment of the original Chromosome 1 (1d: 1-derived) and the Chromosome 4 (4d).
When a Post production bank with cells that had grown in serum-free media for about 20-30 cell cycles was established, a new scenario was found; now only 88% of cells showed the large GoI-chromosome and 12% showed the GoI in a new, smaller chromosome. The emergence of this smaller chromosome can be explained by another translocation occurring between the large GoI-chromosome and a rearranged chromosome consisting of a composite of chromosome 7 and X fragments.
A likely interpretation is that the change in culture condition from serum-containing media to serum-free culture selected for a better growth of cells with the rearranged GoI chromosome. Because a rearranged subpopulation was small initially, less than 1 in 100 cells, it could not be seen in the MCB. Read the full paperPaper details
Authors


Florian M. Wurm and Maria João Wurm
Published in
Biotechnology Journal, 2021, July, Volume 16, Issue 7; doi.org/10.1002/biot.202100165
Introduction
Over the last 3 decades, CHO cells have become the most popular cell line for production of recombinant proteins for human therapy. In fact, nine of the world's 15 top selling drugs are protein therapeutics derived from CHO cells—with 76 Billion US Dollars in sales in 2018 [1], and total sales of CHO products today far exceeding 100 billion US Dollars/year. All CHO cells go back to a poorly described immortalization event that occurred in the late 1950s in an adherent, glass dish-maintained culture of the laboratory of Prof. Theodore T. Puck (1916–2005) [2], the founder and director of the Eleanor Roosevelt Institute for Cancer Research in Denver, Colorado, USA.
CHO cells had not been envisioned for production or even for lab-based protein expression. Their early popularity was due to the ease of investigations on cytogenetics and associated studies on mammalian genes. Scientists liked their large chromosomes, visible under light microscopy. In addition, metabolic mutants of cells could be established [3]. Some of these mutants were found to be correlated with recognized chromosomal rearrangements and thus supported ground-breaking and fundamental studies of genes, their positions on chromosomes and their functions in a mammalian in-vitro system. An example of such work is shown in Figure 1, modified from reference [4]. “Giemsa-bands” allow to identify specific chromosomes (even if similar in size) in mammals and/or in cells derived from them. The figure demonstrates structural changes (inversion of a chromosomal fragment and a translocation). Some of the changes correlate with metabolic functions of the concerned genetic markers. The studies enforced insights gained by Thomas Morgan Hunt (1866–1945) on linked inheritance of genes (synteny) when localized closely to each other on a chromosome.
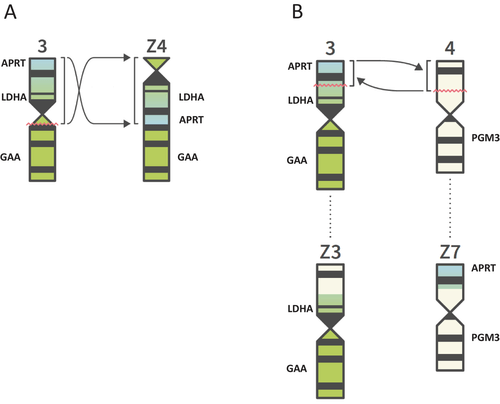
Generation of three Z-chromosomes by rearrangements. Sites of several enzyme encoding genes are indicated. APRT, LDHA, GAA, PGM3 indicate approximate gene locations of corresponding enzymes. The images are re-worked figures from [4], done by C. P. Wurm, Montpellier, France. (A) Inversion of a major chromosomal fragment within chromosome 3 results in the generation of a new, unusual chromosome Z4. Gene locations are changed accordingly and thus follow the prior determined banding patterns. (B) Reciprocal translocation between chromosomes 3 and 4 results in the generation of two new, unusual chromosomes Z3 and Z7. Note: The color shading in the image of Giemsa-banded chromosomes was introduced to better visualize the new arrangement within the emerged Z-chromosomes.
Genomic rearrangements of DNA occur frequently in CHO cells and thus provided an approach to link gene locations with functions. Many cell lines emerged or were selected for specific phenotypes, sometimes after irradiation or chemical mutagenesis and were subsequently named to differentiate them from each other. Some of these names are still in use by the biologics producing industry. Such names (shall) typically indicate the origin of cell populations, but do they provide information on their utility?
This review will refer occasionally to the results of karyotyping, to explain the genetic/genomic evolution of CHO cells. This method is highly suitable and at times more efficient than genome sequencing, to visualize rapidly genomic modifications in both individual cells and in populations of CHO cells.
CHO cells show, in comparison to cancer cells, a relatively stable chromosome number (!) matching globally the diploid Hamster with 22 chromosomes (11 pairs), albeit with considerable structural instability of all chromosomes. The overall genome size of these cells is slightly smaller than that of Chinese Hamster Cricetulus griseus, characterized by haplodiploidy, losses of genes and trends for rearrangements of “normal” Hamster and other chromosomes that have already unusual structures. We quote [3]: “the modal chromosome number is 21, or 20, in the case of one subclone isolated by Kao and Puck (1968), compared with a diploid number of 22 for the Chinese hamster. Although CHO cells carry many chromosomes which differ from those of the diploid Chinese hamster karyotype (Kao and Puck, 1969, the modal chromosome number of a clone is usually constant over many months or years.” The mentioned reference for the Chinese Hamster karyotype is in [5].
A body of literature states that immortalized cells are very similar to cancer cells when comparing their genetic and phenotypic instability, based on an enormous number of genetically diverse cells in a cancer patient or in bioreactors [6-8].
This discussion on cell line utility for expression of recombinant proteins conveys that ancient names provide little information on their genetic constitution and essentially nothing on their phenotypes for industrial use. It is misleading that “old” names indicate usefulness for any specific approach in cloning of genes, in expression yield, or—in the context of industrial application—for culturing these cells in deep tank bioreactors. Even verifying the authenticity of the name is impossible since a documented history of subcultivations cannot be provided.
We will revisit some of the profound studies done with these cells during the 1960–1980. This will allow us to better understand the genomic plasticity and corresponding phenotypes when cells are now grown in fixed-bed culture systems, or in stirred or shaken bioreactors.
CHO origin, immortalization, and names
CHO cells were established 1957 as a cell line. For decades derived cells served as an excellent source for fundamental research in molecular cell genetics. Cell populations were used by laboratories to study gene numbers, structures and locations of mammalian genes, and for the purpose to unravel functions and genetic principles of chromosomes in mammals. A 1963 paper refers to CHO-pro– (minus) cells [9], which appears to be a dominant phenotype of all CHO cells [10]. Thus, the loss of proline synthesis capacity was apparently a very early event, before the distribution and use of derived cell lines. Among the derived cell populations was the “K1” cell line that was claimed to be derived from a single cell—a “clone” [11]. Many other auxotrophic mutants of CHO cells were found and/or generated [12, 13].
The immortalization event of CHO cells, that is, the conversion of the ovary-isolated primary cells into an adherent cell line, occurred sometimes during regular subcultivations in sterilized glass flasks or dishes. At the time, cells were cultured using media containing serum or major fractions of serum, mostly of bovine origin, at concentrations of 10–20%. No chemical or physical exposure appeared related to the immortalization event. It had occurred “spontaneously”. In the 1958 publication,[2] we find “and one (cell line) arising from the ovary of a Chinese Hamster, was selected for long-term cultivation in the complete growth medium of Table I. These (cells) have now been carried for more than 9 months during which they have undergone a minimum of two generations per week, or a total of 78 generations, equivalent to 1023 progeny”.
This remarkable statement demonstrates the robustness and ease of handling of these cells. The 78 generations of the cell population should also be considered with respect to the genetics. 1023 cells, equivalent to 350 million tons (1 cell weighs 3.5 × 10-9 g, the weight of all mankind today being about 450 million tons), are the product of error prone DNA/chromosome duplications! A heterogenic cell population without maintenance of diploidy emerged, even before any specific names were coined. We have referred to these cells as CHOori before.[14]
The K1 cell line was established in the late 1960s by Dr. Fa-Ten Kao, then a Senior Scientist in Puck's laboratory, as a clonally derived cell line, from the earlier mentioned CHO-Pro– [minus] cells.[12] “Mutagenesis experiments were performed with CHO Pro – and its K1 subclone”.[11]
No information is provided how the cloning occurred, but maybe cells were scraped off or aspirated from an adherent colony with a pipette—similar to the ones in the image provided in the Kao and Puck 1967 [10] paper (Figure 2).
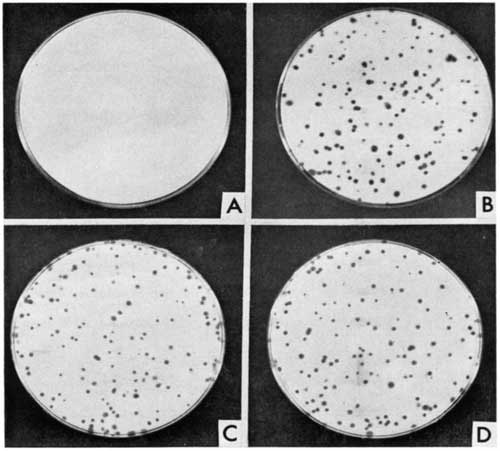
Copy of Figure 1 in,[10] indicating the requirement the CHO Pro– cell line for proline
The “stem number” (The stem number, equivalent to the modal chromosome number, indicates the total number of chromosomes, determined as the majority of cells containing that number, quasi-tetraploid cells are excluded.) of chromosomes in these cells had declined from 22 to 21. CHO Pro– cells were obtained in 1962 by a laboratory in Los Alamos. Analyzing cells by karyotyping, reported on about a decade later (1973), indicated that the modal (or stem) chromosome number was 21 [15]: “Our results demonstrate that only 8 of the 21 chromosomes are normal when compared with Chinese Hamster chromosomes.” Reference is made in this paper to the K1 cell line and its’ similarity to the unnamed cell line's chromosome structures. The K1 karyotype had been published earlier (1970), albeit then with a much lower resolution technique. The K1 cell line was having a modal chromosome number of 20:[11]“ In some experiments, the subclone, CHO/Pro–-K1, which possesses a stemline of only 20 chromosomes, was utilized”. This is in contradiction to a 1985 publication by Puck indicating a modal chromosome number of 21.[12] The 1968 paper by Kao and Puck[11] also refers to the K1 subclone frequently converting to glycine auxotrophy [dependence on the addition of glycine].
The periods between published statements on chromosomal numbers, specific phenotypes and applied names of cell lines are typically long (years) and do not provide culture details for these periods.
We bolded “clonally derived cell line” above: Simply cultivating cells results in drastic genetic changes in populations, clonal or not, and this has been verified frequently by karyotyping. The susceptibility for rapid changes of the genome of individual CHO cells at each mitotic cycle has been discussed in a paper using the term CHO Quasispecies when referring to clonally derived cell lines [16]. The term “clone”, frequently used, perpetuates an impression for non-experts that such cells are genetically identical (or near identical).
Having identified a good CHO “clone” actually means that the population of cells has maintained to a high degree (>70%?) the transcribed DNA of interest. This population may continue to deliver for a few more weeks the product with yields of 60–100% of the initial productivity. In a good “clone” the majority of cells have, thus, kept the chromosome or chromosome fragments containing the gene of interest. The majority of cells—even if diverse genetically—while transcribing the exogenous DNA will also assure growth and overall metabolic robustness. Populations emerging after cloning are related to each other, a CHO Quasispecies. However, the overall genetic diversity of such clonally derived cell populations remains and exceeds the genetic diversity of populations of a biological species.
Most labs, having worked with the early CHO cultures, have been dismantled and/or handed over to successors. Puck, a highly respected researcher with many achievements during his career, has passed away. Prof. Fa-Ten Kao is 86 at the time of writing this text. Our attempts to identify a laboratory that may still have frozen cell vials from the 60–70s have not been successful. Prof. Lawrence Chasin, the originator of the first industry-used cell lines [17], is maybe the only one who has kept cells generated first in the 1970s in his freezers. To replenish his stocks of cells, he repeatedly refroze cells from fresh cultures into small research banks for distribution (personal communication).
The cells are kept in liquid nitrogen and are grown with FBS containing media. Chasin's DHFR-negative CHO cell line, named CHO DXB-11, (also sometimes referred to as CHO-DUK) became the host system for the first CHO produced pharmaceuticals, such as the Genentech Inc. developed Activase Tissue Plasminogen Activator (TPA) or the AMGEN Inc. EPOGEN Erythropoeitin (EPO). To obtain recombinant cells synthetizing these proteins, DHFR-expression vectors were constructed that also contained the DNA encoding human TPA or Erythropoeitin. The functional DHFR gene on the vector would thus “repair” the deficiency in cells and would allow the selection of recombinant CHO cells co-expressing the DNA for TPA or EPO.
Table 1 provides information on some of the cell lines utilised during the period of 1960 to 1990, contributing to knowledge on mammalian genetics, chromosome identity and structures, gene locations, etc.
Table 1. CHO cell lines used in fundamental research from 1957 to 1990
| Name | R&D work for | Period of R&D | Modifications (genetic/metabolic/special exposures, treatments) | Derived from | Comments | Relevant references |
|---|---|---|---|---|---|---|
| CHO-ori (1957) | Cytogenetics, chromosome structure analysis | 1956-1960s (?) | None reported (spont. immortalization) | Primary culture of Ovary tissue (outbred hamster) | Many labs used these cells prior and after K1 became popular | Puck, Ciecuira, Robinson 1958 |
| CHO-Prominus (1957) | Cytogenetics, chromosome structure analysis karyotypes | 1957-1980 | Proline auxothroph | CHO-ori | Numerous derived cell lines were then named CHO Pro– 3, Pro– 5, and so on. | Ham 1963 |
| CHO-K1 (1968) | Mutagenesis, metabolic studies, karyotype analysis | 1968-today | Proline auxothroph | CHO-Prominus | Confusing chromosome Stem number (21/20) Serum-free growth of cells |
Kao, Puck 1967 Deaven, Peterson 1973 Hamilton, Ham, 1977 |
| CHO-S (1974) | Mutagenesis, metabolic stud. | 1974-today | Proline auxothroph Suspension growth Genetics of phenotype variation of suspension culture cells |
CHO-ori | Mendelian inheritance of phenotypes of cells on soft agar. | Thompson, Baker 1974 Konrad et al. 1977 |
| CHO-DX B11 (1980) (also ref. to as CHO-DUK) | DHFR gene structure, metabolic studies, gene amplification, MTX treatment, gene transfer | 1980-today | Proline-, glycine-, hypoxanthine-, thymidine-auxotoph (DHFR minus/DHFR minus) GHT minus |
CHO-K1 after Chemical and gamma-ray radiation |
One transcribed DHFR gene remaining, with single AA mutation CHO-DUK NOT same as DX-B11 |
Chasin, Urlaub 1980 Kaas et al. 2015 Turilova et al. 2021 |
| CHO-DG 44 (1983) | Genomic structure of DHFR sequences Simplified gene transfer with DHFR |
1985-today | Proline-, glycine-, hypoxanthine-, thymidine-auxotoph (DHFR minus/DHFR minus) |
CHO-Pro-3 following treatment w. MTX for gene amplification and Y-rays | Note: the paper by Urlaub et al. mentions DG 41 and DG 42, but not DG 44 | Urlaub et al. 1983 |
Industrially applied CHO cell lines
Names as “CHO-Pro–,“ “CHO-DUK,” “CHO DXB11,” “CHO-K1,” “CHO-DG44,” “CHO-S” falsely imply how to differentiate cells from each other. At the time of generating these names, all cells were grown in the presence of serum and in dishes or flasks. The majority of industrially used CHO cell lines are now grown in chemically defined media in suspension cultures in stirred or shaken bioreactors[18, 19]. However, Prof. Richard G. Ham (1932-2011) grew CHO cells serum-free in 1977, prior to similar efforts in the early biotech industry [20]. Interesting in this context is also L. Thompson's publication, the same year, using the first suspension culture of CHO cells with a Mendelian type inheritance of phenotypes. When plating them as single cells on soft agar he could reproduce two phenotypes, “arrested dome” and “fried egg” morphologies of colonies.[21]
Obtaining highly productive recombinant cell populations in large-scale bioreactors is a complex task. Transfection with optimized expression vectors for expression of the transgene(s) from strong promoters leads to the selection of clonally derived cell populations with genome-integrated vector DNA. Frequently now antibiotics are added to the culture medium, with the corresponding resistance marker on the vector. This approach can replace the above mentioned DHFR- or Glutamine Synthetase (GS) selections. When using antibiotics for selection any CHO line can be used.
Productivity
The most widely used industrial principles for CHO-based manufacturing under suspension culture were developed in South San Francisco at Genentech Inc., USA. Adherent cells could not produce sufficient amounts of human Tissue Plasminogen Activator (TPA), for a successful introduction into the US-American market. The expected dose of 10 mg/patient was found in clinical trials to be 10 times higher, resulting in a critical manufacturability problem. The only solution was to have the producer cell line grow as a suspension culture in large stirred bioreactors (STRs). Drawing from insights gained with the BHK cells for Foot and Mouth Disease Virus production [22, 23], the conversion to suspension cell populations for STR based manufacturing was eventually done—with cells derived from a bank of cells under adherent culture. The term “adaptation” was used for the process towards suspension culture– wrongly as we would argue. (This seems academic, but a selection of genetically different subpopulations with a higher capacity to grow in suspension culture is the most likely explanation, that is, the change in phenotype is connected to a suspected modification of genotype overall.) The process that delivered the required product quantity for the US-American market was initially based on the use of 2000 L and then 10′000 L bioreactors and had a volumetric yield of 50 mg L−1. From today's perspective, this is a low yield, but one needs to remember that the optimization of cell culture media compositions was just starting at the time and a cell density of 2–3 × 106 cells mL−1 was considered excellent for a 7-day batch process. The production line was derived from the aforementioned “CHO-DXB11” cell, which conveniently allowed transfer of the DNA of interest into the CHO genome by co-transfer with a DHFR expression cassette. Dr. Chasin had established that one allele of the functional DHFR gene was missing in his cell line (by loss of a chromosome fragment), whereas the other had a single nucleotide mutation [17]. To increase productivity methotrexate (MTX)—an antagonist to DHFR—was used. This approach, called gene amplification selects for populations of cells that have increased the copy-number of the DHFR/TPA vector within CHO chromosomes. The mechanisms for such gene amplifications have been studied in cancers and cancer-derived cell lines, as well as in CHO cell lines. They involve drastic chromosomal rearrangements. In other words, the known genomic malleability of CHO cells was used to select for subpopulations of cells, emerging spontaneously, to increase productivity [24, 25].
In optimized fed-batch processes, CHO cells can produce today 3–10 g L−1 of recombinant antibodies and other molecules, with chemically defined media that maximize viable cell densities. These processes apply animal component free media. Research into medium formulations provided much enriched compositions. Medium formulations are non-published intellectual properties. High yields have been shown without any host cell engineering [18, 26].
Cloning, subcultivations of cells, genetic bottlenecks, and population dynamics
Regulators request evidence of clonality when producing a therapeutic protein—the reasons being debated in the industry [27]. The topic was discussed again recently by scientists of the Food and Drug Administration [28]. Importance of clonal derivation was maintained, while admitting that “a master cell bank created under even the strictest conditions possible may never be truly “clonal””. The word “may” is misleading—the MCB cells will never be clonal in the sense of its typical use in biology. FW [16] recommended minimizing growth-restricting conditions. But this was considered by the FDA authors “incompatible with the nature of modern biopharmaceutical production”. We do not understand this: Growth-restricting environments can be avoided, or at least reduced. This supports maintenance of an averaged phenotype of even diverse but genetically related populations.
Clonality is hoped to increase robustness/reliability or reproducibility of the manufacturing of the product, such as the avoidance of a second cell with a different genetic background to be the “unknown cause” of a mixed and/or unstable population. Insistence of clonal derivation was not enforced during the 80s and 90s. FW has picked clonal populations as visually marked colonies of hundreds of adherent cells from Petri dishes using sterilized cotton swaps or “cloning rings.” Cells adhering to the cotton or being aspired off were transferred into the medium within a micro-well. Colonies on a plate could be easily compared in size. Cloning single cells, a standard today, was done then with efficiencies below 10%, even with fetal bovine serum as part of the medium composition.
Several “cotton-swab” derived cell lines are the source of globally marketed products still today. The relative genetic and production homogeneity of emerged populations and their overall production stability was mostly based on the purified protein product, assuring that the manufacturing process delivered a reliable and reproducible pharmaceutical [27].
Whatever the starting point of a cell population for production, likely a suspension of frozen cells in a small vial, cells are exposed to many steps. All work is also associated with the generation of Research Banks, Master Cell Banks and Working Cell Banks, derived from short-term, expanded cultures to be aliquoted into small containers, medium exchanged and exposure to freeze-thaw cycles.
To estimate the number of these frequently critical cell processing steps is impossible to judge. Again, cloning does not protect against population heterogeneity. This will become more transparent in the next chapter.
Genetic and phenotype implications of non-clonal and clonally derived cell populations
It is likely that 200 or more subcultivations have been executed since the cells were named during the 1960–1980 period. We wish to put this into a calculation that starts with 2 × 106 cells. With only 100 subcultivations the astonishing number of 1 × 1096 cells could have been generated. The necessary DNA replication of a 3 billion base pair genome will involve numerous modifications of the underlying CHO genomes. The “normal” mutation rate is 10–8 per locus/cell in mammalian systems [29]. In cultures of a suspension mouse cell line, a mutation rate of 1–7 × 10–7/cell/generation (generation = cell duplication) was found [30]. With these numbers in mind, and the large number of duplications in regular maintenance of cell populations occurring, CHO cells will accumulate very fast mutations that will affect phenotypes. Mutation driven selection will lead to evolution of cell populations, depending on the various environmental conditions—following the concepts elegantly and convincingly described by Charles Darwin,[31] more than 150 years ago. Genome and phenotype affecting changes will be even more striking when cloning steps are involved that generate “genetic bottlenecks” in which a single cell gives rise to an emerging new population.
Complex cytogenetics of cloned and non-cloned cell populations
T. C. Hsu [1917–2003] published 1961 a landmark review article “Chromosomal Evolution in Cell Populations” [32] in which he stated that “cells grown in-vitro change from their original genetic composition to a heteroploid condition”. Recent (2021) karyotyping of CHO cells verified this statement, while drawing also important insights from karyotyping of CHO cells over decades [33]. Next generation genome DNA sequencing techniques on cancer cells and other immortalized cell lines revealed that some genomic changes can be dramatic, even “catastrophic”, resulting in breaks into multiple fragments of chromosomes (chromothripsis). However, the majority of chromosome modifications in many cancer cells and cell lines result in “simple” translocations, fragment inversions, multiplications, losses of fragments and entire chromosomes, losses of heterozygosity, polyploidizations. HeLa cells may be an extreme example: These cells, the first established cell line in history, show these modifications, including chromothripsis. More than 60′000 papers have been published using HeLa cells [34]. The cells were obtained and established as a culture by the treating doctor, G. O. Gey (1899–1970) of Mrs. Henrietta Lacks. She passed away in 1951 in Baltimore at the age 31 due to complications from a malignant cervix carcinoma that had invaded the entire body and resulted in an acute kidney failure [35]. The cells were instrumental in establishing cell culture technologies as they are used still today and with tremendous benefits for biology and medicine. A recent genome sequence analysis [36, 37] of HeLa populations showed hyper-triploidy on average, with some genomic fragments having a ploidy of four or higher and presenting 20 highly aberrant and rearranged chromosomes, four of which having been the subject of chromothripsis. Also, the HeLa genome exhibits significant stretches of homozygosity, explainable only by losses of the heterozygote partner chromosomes or fragments thereof. In addition, there is a plethora of single nucleotide variants, indels (“short” DNA- fragment—50 base pair to 10′000 base pair—deletions and insertions) and copy-number changes, including more than 2800 structural variations, dominated by large deletion events. The numbers are staggering—approximately 4.5 million single nucleotide variations and about 500′000 indels!
A genome sequence of CHO cells was published 2011: “The genomic sequence of the Chinese hamster ovary [CHO]-K1 cell line” [38]. We have serious reservation for the dual use of “the” in the title of that paper. The article seems to imply that there is such a thing as one genomic sequence and one CHO-K1 cell line. In the article the term “ancestral” was used, probably referring to the fact that the cells were grown in simple culture media with 10% Fetal Bovine Serum (FBS) and were obtained from the American Type Culture Collections (ATCC).
The published genome sequence represented 21 scaffolds as non-interacting DNA blocks, interpreted as 21 chromosomes. An image of size-ranked chromosomes was shown. As a reminder, the 1973 karyotype paper [15] showed also a distribution of chromosome numbers per cell. They ranged from 19 to 44 [1 pseudo tetraploid cell], when analyzing 50 metaphase spreads. Thirty eight of these show the chromosome stem number of 21, six cells have 20 chromosomes and four have 22! The CHO K1 genome sequence paper does not provide chromosome-based sequence data and gives little information on differences from the sequence composition of the 11 chromosome identities of the Hamster. The paper did not comment on indels and single nucleotide variations, nor does it discuss the level of ploidy and homo- and heterozygosity. Also, the presence of endogenous retroviral sequences—a major concern for products made with immortalized cells—giving rise to A- and C-type retrovirus like particles is not mentioned. The relative scarcity of such particles in CHO cells and the mostly non-functionality of corresponding sequences in their genome [39], in comparison to mouse-derived cell hosts, was one of the reasons why CHO cells were considered a safe(r) host system for human therapeutic protein production.
Highly useful insights gained by karyotyping of a CHO DXB-11 cell line and comparison with other CHO karyotypes were just recently published [33]. In this cell line, delivered to Russian Cell Culture Collection in 1984, only 17% of cells appear to have a karyotype that would be accepted as the main structural variant, that is, repeatedly occurring structural (chromosome) features in cells. This main structural variant [MSV] shows six normal hamster chromosomes (pairs of the Hamster chromosomes 1 and 5, and a single representative of the Hamster chromosomes 8 and 9) and 14 structurally rearranged chromosomes (i.e., their origin can be deducted from the banding pattern), including four marker chromosomes (see Figure 3). Eighty three percent of cells—based on an analysis of 112 karyotypes—displayed clonal and nonclonal additional structural rearrangements of chromosomes. The authors also reveal that a prior assumed equivalence of CHO DUK and CHO DXB-11 cannot be verified. Apparently, a naming confusion occurred, since the here shown karyotype differs from the published CHO DUK karyotype. This example of an “old” cell line, analyzed recently, shows quite strikingly how misguided identity assignments can be and how relatively small the numbers of cells can be in a population that appear to be characterizable by a defined genotype.
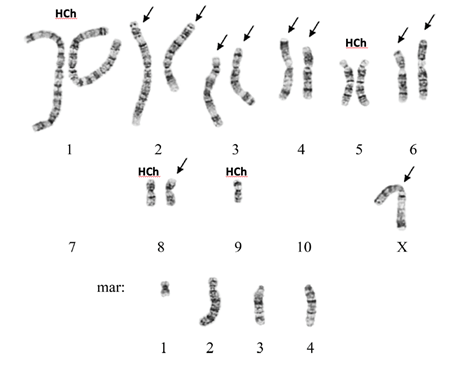
G-banded karyotype of the Chinese hamster ovary DXB-11 cell line, with its main structural variant. Chromosomes indicated with arrow show a deviation from the original hamster chromosomes but can be still assigned to the original chromosome from which the majority of its’ DNA is derived. The mar (marker) chromosomes do not show a banding pattern that allows to assign them to original Hamster chromosomes. Hch indicate “normal” hamster chromosomes (image reproduced and modified with the authors permission, [34]).
Recombinant CHO cells used in pharmaceutical manufacture can show similarly confusing complexities when considering their karyotypes [40]. The Merck (Darmstadt) group used chromosome painting techniques to identify origin and structures of the chromosomes of a cloned (!) cell line producing a therapeutic protein. The producer cell line was initially grown with FBS under adherent culture and a Master Cell bank was generated. A fixed-bed bioreactor was used to manufacture a biopharmaceutical. Eventually the company decided to “adapt” the cell line of FBS -free processes. Strikingly, cells derived from an extended cell bank (ECB) made with cells from a Post-Production Cell bank (PPCB) showed a 12% subpopulation with a different site of integrated gene-of-interest DNA than originally found in the FBS-grown cells. (The cell line was generated using the DHFR/Methotrexate gene amplification approach.) The Fluorescence-In-Situ-Hybridizations (FISH) of cells in Master- and all Working Cell Banks had the DNA of interest as part of the largest chromosome structure found in most metaphases (likely a Hamster chromosome 1 derived). Now, in the ECB 12% of cells showed this DNA in a much smaller chromosome. Numerous other cytogenetic modifications were also observed. Only nine additional population doublings (= generations) occurred from the FBS containing MCB to the serum-free Working Cell Bank (sfWCB). At the WCB level all seemed ok. Yet, some ECB cells had the DNA of interest translocated and/or otherwise rearranged as part of a mosaic chromosome containing fragments of chromosome 1, 7, 4, and X! Between WCB cells and ECB cells cultures less than 50 days elapsed, of which a 38-day period involved a production run that also included a temperature shift to 31°C. It is fair to assume that at least 30 cell population doublings had occurred.
Interpretation? The modification of the culture condition—from serum-containing to serum-free media—resulted in a selective condition that favored the growth of a cell or a subpopulation of cells (less than 1% of the total population) carrying a chromosome resulting from several rearrangements. We cannot know how and when the cytogenetic modifications occurred. It could have been present in the very first MCB population or even shortly after cloning of the cells that gave rise to a cell population for later use. However, this modification was not captured by even the most sophisticated FISH technology, if the frequency of cells carrying this type of cytogenetic modification is below the detection limit: Usually 100 metaphase cells or less are typically analyzed by FISH. With the chromosome painting technique several additional cytogenetic modifications were observed in other chromosomes than the new chimeric structure. How these cytogenetic changes, including the one containing the recombinant DNA, contributed to the selective growth advantage is a mystery. However, the cell population established in the Master Cell Bank and subsequent Working Cell Banks must have contained these. The visualized cytogenetic modification is just “the tip of the iceberg.” We must assume that other more obscure genetic changes will occur in CHO cells, such as the single nucleotide variations and the “short” DNA fragment indels, referred to in the discussion of the HeLa cell genome above. While genome sequencing has provided fundamental insights in biological sciences with higher organisms that have multiple layers of control over DNA-replication errors and that assure functional species-defining chromosome structures, it is quite striking how little massive data accumulation can provide when dealing with poorly controlled genetic systems, as faced in cancer and with immortalized cells.
An insightful publication by Nicole Borth and her team in Vienna [41, 42] describes the evolution of karyotypes as well as the overall distribution of modal chromosome numbers in CHO cells over a period of 6 months. The study addresses cells as CHO-S, CHO-K1 and CHO-K1 8 mM glutamine. A CHO-S cell line was derived from the company Thermofisher (Waltham MA, USA). The CHO-K1 was obtained from ECACC (European Collection of Authenticated Cell Cultures, Porton Down, UK), “adapted” to grow in protein free medium CD-CHO [Gibco], and the CHO-K1—8 mM Gln, was further selected for growth in the same CD-CHO medium without the addition of glutamine and was then called CHO-K1 0 mM Gln.
The analysis showed relative stability of the modal chromosome numbers of these cell lines—the CHO-K1 derived cell lines at 19 and 18 (!), the CHO-S derived cell line at 22. All three lines showed a large variation of chromosome numbers per cell, including near tetraploids and cells with lower chromosome numbers. The authors also compared an A with a duplicate B culture and found small but significant trend differences. In one of the CHO-K1 derived cell lines the differences between the A and B cultures were becoming more pronounced!
The karyotypes of all cell lines were changing, with translocations, chromosome fragment losses, and other major modifications. The authors write: “No single chromosome was not also found in a rearranged form.” They describe certain patterns for chromosomal rearrangements over time and they point to the possibility to identify few “most stable” chromosome structures, among them at least one copy or a major part of the Hamster chromosome 1.
The above mentioned cytogenetic changes over a period of 6 months occurred without any apparent selective pressure and/or the application of population bottlenecks. Another striking quote is “….the occurrence of chromosomal aberrations is increasing with time in culture, genomic instability is an unpredictable and uncontrollable property and no population is stable or uniform over a longer period of time.” (bolded by the authors of this article). This statement sounds very similar to the one phrased by Dr. Hsu, 60 years ago [32].
In conclusion, readers will understand now that CHO cells cannot be defined under the rules of the more stringent genetics of animal and plants species. In CHO cells, evolution of genomes is a constant phenomenon and at times, with drastic results and occurs within time frames of typical laboratory cultures. It seems that the success of CHO cells in manufacturing may be at least in part a result of the capacity of CHO populations to approach and achieve fitness for bioreactor environments relatively fast.
Cloned cell populations for pharmaceutical protein production—How to arrive at and keep a relatively stable phenotype?
The term “adaptation” is frequently used in the industry for cells that will be switched from one cell culture medium to another one, or for achieving suspension culture in serum-free media. With the insights discussed above, the term selection is more suitable. Due to the large diversity of genotypes, spontaneously arising in any cloned or non-cloned population, there are apparently sufficiently diverse individual phenotypes within a given cell population to overcome growth limitations which can be sometimes observed. This can occur fast: 5 to 10 subcultivations -15–40 population doublings—may be sufficient. We have seen such selective fitness emerging to a new formulation frequently in our work and have noted minimal effects on overall viability and/or only transient declines in maximal cell density until a full recovery.
Another observation seen in about 10% of clonally derived cell populations is a shift in maximal cell density which occurs after 10–20 subcultivations with cells under stability studies. This shift to a higher maximal cell density—a phenotypic parameter of high importance upon scale-up- can occur over a period of 4–7 weeks. An example is the following: Cells from a freshly thawed cell bank were growing well to 10–13 × 106 cells mL−1 in 3–4 day culture, tested and measured over 3–4 weeks. However, then over a period of 2–3 further subcultivations, the maximal density “jumps”up to 15–17 × 106 cells mL−1 and then remains constant over the remaining study period (2.5 months). We did not see a declining viability at any time (unpublished). We interpret this as a selective fitness improvement.
A manufacturing cycle for recombinant protein pharmaceuticals starts with 1–2 mL of cells in a frozen vial from a Master- or Working Cell Bank. Large stirred bioreactors (1000–20,000 L) deliver in the end the non-purified product towards down-stream processing. The time required to go from a vial to production vessel will typically take 3–5 weeks, with another 1–2 weeks required for the production phase. Since several productions may be necessary from one maintained seed culture, the overall production campaign executed from a single vial can last 3–6 months. Production stability tests need to cover this period. In our labs, stability studies are executed at least with 3, preferably 5–10 clonally derived cell populations—from Research Cell Banks. A final decision on the production cell line and the thus to be established MCB is done after comparing data from the stability study.
Stability studies determine, in weekly intervals, the quantity of product produced by the clonally derived cell population for a period of three or eventually 6 months (>80 generations). One can also study growth rates, maximal cell density and quality of the product. A cell line is considered stable when a possible decline in productivity over the observation period is less than 15%.
Obviously, different gene-transfer and selection approaches affect stability to a different degree. We cannot discuss these here. However, growing clonally derived cell populations under controlled conditions over more than 3 decades have provided us and regulators with satisfying results to provide reliable cell populations for pharmaceutical manufacture. Why does this work? Population genetics [44, 45] provides a reasonable answer, and here are a few lines of interpretation.
Cell banks consist of cells with a diversity of genomic structures, many of them visible by cytogenetic studies. One could study and establish the larger, identifiable cytogenetic diversities for each cell bank. This would be interesting when done with many such banks of the same and different products from one host system. Such studies could lead to deeper insights into a potential “core” karyotype of CHO cells cultured under defined conditions and transfected with different product genes. This is a labor-intensive exercise which apparently no academic or industrial laboratory would consider worthwhile and it is not really useful for individual cell banks.
Full genome sequencing is of little use here either. With 10 × 106 cells or so in a vial we must assume hundreds of major and minor structural chromosomal modifications, different from the one karyotype and one genome sequence in the cloned cell. The question will be at what time (after/before banking) and how frequently such sequencing should be done. Also, it matters that all sequencing will “average out” obtained data over thousands of cells. Is the “the 1000 human genome project, started in 2008 a good model? Would a 1000 CHO genome project provide relevant and useful data? We are doubtful. We simply do not know enough about the impact of CHO genome structures/sequences and the observed phenotypic differences, particularly when the observations we make are again the “average” phenotype of populations.
Individual cells are likely to exhibit different phenotypes when expanded. However, evidence support the notion that clonally derived and non-clonal cell populations contain a Main Structural (karyotype) Variant (MSV), detectable in a large(r) percentage of cells [12, 24, 33, 34, 41-43]. Such cells should also exhibit phenotypes closer to each other than do cells that had undergone massive genomic rearrangements. A Gaussian distribution with a smaller or larger peak of genomes around the MSV is likely. Variations away from this MSV would emerge permanently and sporadically. However, MSV carrying cells can maintain this genotype/phenotype and thus assure the progression of this MSV of the population. Figure 3 is an example of such a MSV of which only six chromosomes seem equivalent to the originals in the hamster.
Several authors have pointed towards certain chromosomes of lines that seem to be less likely object of structural modifications. Turilova et al. [33] refer to several “most stable” chromosomes in their cultures of DXB-11 cells, one being the single “authentic” chromosome 9, and other chromosomes that were identified as derived from the X-chromosome and from the chromosomes 2, 4 and 6. Interestingly, these latter structures represent rearranged chromosomes, clearly different from the original hamster homolog. Thus, it is fair to assume that certain authentic and non-authentic hamster chromosomes in a population have a (slightly?) higher probability to be maintained as part of a MSV. However, it would be too simplified to conclude that authentic hamster chromosomes have a higher probability for being maintained unaltered, as indicated by publications of the Borth group in Vienna [41,42].
The observed phenotype of a given cell population is the result of all genotypes. Among this, one should not underestimate the role of short-lived cells that emerge and die in a population in this context. Even in a “100% viable” culture, cells die permanently—we just do not see them in cell counting methods. These cells deliver into the medium components that may support the growth of others. Other strange phenomena occur, observed in hybridoma cell cultures. One example is this: A larger than typical cell engages into cell division, resulting “almost” in two daughter cells. This is shown in a time-lapsed video recording of cells under a microscope. However, within 45 min, further separations into 5 cell-like structures occur, to then fuse back within 40 min first to three and then to two separate cells, [46] see also [14]. One wonders what may have happened to the genome of the first cell during the subsequent events.
Thus, genetic and phenotypic stability of a population of cells is to be understood as averaged from diversity. If there are enough cells in the population that are, in terms of their physiology, a best fit to the environment to which they are exposed to, then this phenotype could turn out to be relatively stable, as long as this environment is kept. An interesting paper provided opportunities of the diversity of clonally derived cell populations for the generation of recombinant proteins that require specific metabolic activities for obtaining acceptable quality of the protein of interest. This work demonstrates the utility of leveraging diversity toward delivering specific performance characteristics of cells [47]. Heterogeneity in suspension cultures of CHO-K1 cells was revealed by single-cell transcriptome analyses, indicating diversity due to epigenetic influences, but also revealed mitochondrial genome variation and heteroplasmy in cells [48].
A note on DHFR/methotrexate (MTX) based cell lines use in industry: in the early years of our industry, the DHFR/MTX approach to high yielding CHO cell lines was dominant,[24] later joined by a similar approach using glutamate synthetase (GS) and methylsulfoximine (MSX).[49] Other selective agents, such as antibiotics, are also used today, including their corresponding selective markers that convey resistance after transfer with Zn-finger nucleases [50, 51] and CRISPR-Cas mediated gene transfers [52]. Together with modified expression vector cassettes and additional elements in plasmids, these approaches enhance targeting the gene of interest sequences into open chromatin. However, with single genome targets into specific chromosomal sites, one should take the high probability of modified genomes by rearrangements into consideration.
Naming of CHO cells — recommendations and final remark
Clonal or not, CHO cells have such a high-level propensity towards larger cytogenetic and other less detectable genetic changes that a name given 30–50 years ago makes little sense. This is even more so when there is no documented history available that traces back the various modes of culture and media used for a given cell line. While we are fully aware that a widely accepted nomenclature is very difficult to change, we wish to initiate at least a discussion on this topic. Such naming modification—at least for the cell lines used for industrial purposes—would make their use, their analysis by modern proteomics, transcriptomics and genomics and their improvement by targeted and selected modifications much more efficient. We recommend therefor establishing names of cells based on definable and relevant phenotypic features. Well established methods, such as DNA sequencing with properly designed primers and/or other methods, such as iso-enzyme analysis [53] allows to assure a given cell line is in fact CHO derived.
Thus, the basic name “CHO” is entirely sufficient. A suspension CHO cell line grown in chemically defined medium should, when passed on to another user for industrial or research purposes, have this phenotypic description somehow captured in its’ name. There is nothing wrong to identify the company or laboratory which has done most of the work. It seems that some cell lines established by commercial companies (“Lonza cell line”) have at least in part embraced this idea. If genetic modifications are involved (“knock-in, knock-out”) they should be part of it. Examples could be “Lonza CHO CDM GSminus 2002″ “Horizon CHO (animal component-free) ACF DHFRminus 2010″. The chemically defined medium (CDM) can refer to the specific medium applied, and the year indicates the timing of the deposition of a bank. The number of subcultivations under use of the mentioned medium would be useful as well: “Lonza CHO ProCHO523 2002″.
Realistically, we are fully aware that these or any other suggestions will take time to become possibly accepted by the interested CHO manufacturing community. ESACT (European Society of Animal Cell Technology) or the Cell Culture Engineering meetings (USA) could be fora for such discussions. Also, a discussion should be initiated with the ATTC or ECACC. If a new naming standard could be implemented it would provide more clarity, both on the actual derivation of cells and their probability to be useful for protein manufacturing.
In concluding: No text written in early in 2021 can capture all historic and technical details that gives a complete and profound understanding on where the cells in our labs came from. We have dealt with them cumulatively now for more than 60 years and remain surprised how little we know. We are also impressed how efficient these cells can be modulated in their behavior in bioreactors if one takes the time and studies their physiology under production conditions carefully.
We hoped to bring a bit more clarity to the very obscure history of these cells and also highlight those points and perceptions that are clearly misleading. And thus, whether or not one assumes that the cells in my lab are “K1” derived or “S” derived, does not matter really. What really counts is the handling of cells under well-defined conditions and observing the spectrum of opportunities these cells and their unique subpopulations provide. CHO cells have taken a dominating lead in manufacturing of high-value protein therapeutics in bioreactors and it is doubtful that any other host system will ever achieve the productivities and product qualities seen with these cells, providing products of unsurpassed purity and safety for millions of patients.
Author contributions
(CASRAI CRediT taxonomy) Authors’ contribution(s) to the submitted manuscript are attributed as follows: Maria Wurm, conceptualization-equal, investigation-equal, supervision-equal, writing-, original draft-equal, Florian Wurm, conceptualization-equal, investigation-equal, supervision-equal, writing-original draft-equal.
About the authors
Maria J. Wurm (56), formerly Maria João De Jesus, graduated with a degree in Environmental Engineering from the University of Lisbon and obtained her PhD at the Swiss Federal Institute of Technology in Lausanne (EPFL), Switzerland (1995). Subsequently, she joined the Laboratory of Cellular Biotechnology as a senior researcher. During her tenure at the EPFL, Maria invented several innovative approaches to the suspension culture of animal cells and how to improve their performance in protein production and in gene transfer. Some of these inventions are now widely used in the industry. In 2002, Maria joined as a first and co-founding employee the company ExcellGene SA, Monthey, Switzerland. She hired and trained a high-quality team and managed all activities related to the manufacturing of recombinant protein and virus-vector-based pharmaceuticals made in animal cells in bioreactors. With increasing responsibilities and growth of the company, Maria joined the Board of Directors of ExcellGene in 2006. Maria was promoted to Chief Operating Officer in 2009 and accepted the position of CEO in 2017. Maria is a frequent invitee to international conferences to speak on process sciences with mammalian cells and has published over 40 highly referenced papers.
Florian M. Wurm, 70, trained as Biologist/Molecular Geneticist (University of Giessen, PhD 1980). His career involved academic and industrial work: Behringwerke AG, Marburg (1981–1984), Harvard Medical School, Boston (1984–1985), Genentech Inc, San Francisco (1986–1995), Swiss Federal Institute of Technology, Lausanne (EPFL), (1995–2015), ExcellGene SA, Monthey, Suisse (2001-today). He contributed to the generation of several high-value therapeutic products (cumulatively now sold for multi-billion Dollars/year globally), such as Herceptin®–an anti-breast cancer antibody, Pulmozyme®-for cystic fibrosis and Tenecteplase®–a highly potent thrombolytic agent. His research activities covered Process Sciences, from DNA to large-scale manufacturing, using recombinant animal cells (CHO, HEK and others) in bioreactors. He is/was the founder (2001), CSO (2002-today, CEO (2015–2016), and CSO/Chairman (since 2017) of ExcellGene SA, a family-owned service company for the Pharmaceutical Industry. He retired from the EPFL in 2015. Florian is member and was Chairman of the European Society of Animal Cell Technology (ESACT). He has published, with co-workers, more than 230 highly referenced papers and filed more than 30 patents covering aspects of expression and manufacture of clinical proteins using mammalian cells in bioreactors.
Dedication
To the memory of Professor Fritz Anders (1919-1999), genetics teacher of FW, an early pioneer in oncogene- and tumor suppressor-gene biology at the University of Giessen, Germany.
Funding
ExcellGene funded internal research and provided materials cost free to academic partners. The work of V.A. was partially funded by a post-doctoral grant from the Swiss Secretary of State for Research, Education and Innovation.
Acknowledgments
The authors thank Drs. Victoria Turilova and Tatiana Yakovleva, as well as Dr. Joeri Kint, for reviewing of the paper and for useful suggestions for improvements on interpretation of discussed data available in the literature.
Conflicts of interest
The authors are founders and managers of ExcellGene SA, a service company for the Pharma and Biotech Industry. Both have had long careers in academic institutions and the observations and conclusions made on CHO cells are derived from both employment activities over a period of 40 (FW) and 25 years (MW), respectively.
References
- Top drugs and pharmaceutical companies of 2018 by revenues 2018 Google Scholar
- Genetics of somatic mammalian cells. 1958 Journal of Experimental Medicine, 108(6), 945– 956. | Google Scholar | CrossRef | PubMed
- Isolation of mutants of cultured mammalian cells. 1974 Methods Cell Biology, 6, 209– 281. | Google Scholar | CrossRef
- The genetic map of the Chinese hamster and the genetic consequences of chromosomal rearrangements in CHO cells. 1985 In: Gottesman, M. M. [ed] Molecular Cell Genetics, John Wiley and Sons. 95– 135. | Google Scholar
- Genetics of somatic mammalian cells. IX. Quantitation of mutagenesis by physical and chemical agents. 1969 Journal of Cellular Physiology, 74(3), 245– 258. | Google Scholar | PubMed
- Genomic instability and cancer. 2014 Journal of Carcinogenesis Mutagenesis, 5, 1000165. | Google Scholar | CrossRef | PubMed
- Genomic instability—An evolving hallmark of cancer. 2010 Nature Review Molecular Cell Biology, 11, 220– 228. [PubMed: 20177397.] | Google Scholar | CrossRef | PubMed
- The immune biology of microsatellite-unstable cancer. 2016 Trends in Cancer 2(3), 121– 133. | Google Scholar | CrossRef | PubMed
- An improved nutrient solution for diploid Chinese Hamster and human cell lines. 1963 Experimental Cell Research, 29, 515– 526. | Google Scholar | CrossRef | PubMed
- Genetics of somatic mammalian cells. iv. Properties of Chinese hamster cell mutants with respect to the requirement for proline. 1967 Genetics 55, 513– 524. | Google Scholar | CrossRef | PubMed
- Genetics of somatic mammalian cells, VII. Induction and isolation of nutritional mutants in Chinese hamster cells. 1968 Proceedings of The National Academy of Sciences USA, 60(4), 1275– 1281. | Google Scholar | CrossRef | PubMed
- Development of the Chinese Hamster Ovary cell. 1985 n: Gottesman, M.M. [ed] Molecular Cell Genetics, John Wiley and Sons. 37– 64. | Google Scholar
- Reversion of a Chinese Hamster cell auxotrophic mutant. 1974 Proceedings of the National Academy of Sciences, 71(3), 718– 722. | Google Scholar | CrossRef | PubMed
- Cloning of CHO cells, productivity and genetic stability—A discussion. 2017 Processes 5[2] 20. | Google Scholar
- The chromosomes of CHO, an aneuploid Chinese hamster cell line: G-band, C-band, and autoradiographic analyses. 1973 Chromosoma, 41, 129– 144. | Google Scholar | CrossRef | PubMed
- CHO quasi-species—Implication for manufacturing processes. 2013 Processes, 1, 296– 311. | Google Scholar
- Isolation of Chinese Hamster cell mutants lacking dihydrofolate reductase activity. 1980 Proceedings of The National Academy of Science, 77, 4216– 4220. | Google Scholar | CrossRef | PubMed
- Production of recombinant protein therapeutics in cultivated mammalian cells. 2004 Nature Biotechnology, 22(11), 1393– 1398. | Google Scholar | CrossRef | PubMed
- Large-scale cell culture in biotechnology. 1986 Science 232(4756) 1390– 1395. | Google Scholar | CrossRef | PubMed
- Clonal growth of Chinese hamster cell lines in protein-free media. 1977 In Vitro, 13, 537– 547.| Google Scholar | CrossRef | PubMed
- Clonal variation in colony morphology and growth of CHO cells cultured on agar. 1977 Cell, 10, 305– 312. | Google Scholar | CrossRef | PubMed
- Growth of a cloned strain of hamster kidney cells in suspended cultures and their susceptibility to the virus of foot-and-mouth disease. 1962 Nature, 195, 1163– 1164. | Google Scholar | CrossRef | PubMed
- Production of foot-and-mouth disease virus antigen from bhk21 clone 13 cells grown and infected in deep suspension cultures. 1965 Nature, 205, 1135– 1136. | Google Scholar | CrossRef | PubMed
- Plasmid integration, amplification and cytogenetics in CHO cells: Questions and comments. 1994 Biologicals, 22, 95– 102.| Google Scholar | CrossRef | PubMed
- Effects of methotrexate on transfected DNA stability in mammalian cells. 1990 Molecular Cell Biology, 10(1), 401– 404. | Google Scholar | CrossRef | PubMed
- Maximizing productivity of CHO cell-based fed-batch culture using chemically defined media conditions and typical manufacturing equipment. 2010 Biotechnology Progress, 26, 1400– 1410. | Google Scholar | PubMed
- Industry view on the relative importance of “clonality” of biopharmaceutical-producing cell lines. 2016 Biologicals, 44, 117– 122. | Google Scholar | CrossRef | PubMed
- Considering “clonality”: A regulatory perspective on the importance of the clonal derivation of mammalian cell banks in biopharmaceutical development. 2019 Biologicals, 62, 16– 21. | Google Scholar | CrossRef | PubMed
- The mutation rate and cancer. 1996 Proceedings of the National Academy of Science of the USA, 93, 14800– 14803. | Google Scholar | CrossRef | PubMed
- How variable is an spontaneous mutation rate in cultured mammalian cells? 1993 Mutation Research, 307, 121– 129. | Google Scholar | CrossRef
- On the Origin of Species. John Murray, London, Albemarle Street 1859. 1854 Google Scholar
- Chromosomal evolution in cell populations International Review of Cytology, 1961 12, 69– 161. | Google Scholar
- Chinese hamster ovary cell line DXB-11: Chromosomal instability and karyotype heterogeneity. 2021 Molecular Cytogenetics, 14(1), 11. | Google Scholar | CrossRef | PubMed
- Reflection on Henrietta lacks? Legacy. 2013 Journal of Biosafety & Health Education, 1, 106. | Google Scholar | CrossRef
- The Immortal Life of Henrietta Lacks. 2010 Crown Publishing Group. | Google Scholar
- The genomic and transcriptomic landscape of a HeLa cell line. 2013 G3 Genes Genomes Genetics, 3, 1213– 1224. | Google Scholar | CrossRef
- The fractured genome of HeLa cells. 2013 Genome Biology, 14, 111. | Google Scholar | CrossRef | PubMed
- The genomic sequence of the Chinese hamster ovary (CHO)-K1 cell line. 2011 Nature Biotechnology, 29, 735– 741. | Google Scholar | CrossRef | PubMed
- Presence and transcription of intracisternal A-particle-related sequences in CHO cells. 1990 Journal of Virology, 64(5), 2021– 2032. | Google Scholar | CrossRef | PubMed
- Reciprocal translocation observed in end-of-production cells of a commercial CHO-Based Process. 2015 Journal of Pharmaceutical Science and Technology, 69(4), 540– 552. | Google Scholar
- . Karyotype variation of CHO host cell lines over time in culture characterized by chromosome counting and chromosome painting. 2017 Biotechnology Bioengineering, 115(1), 165– 173. | Google Scholar
- evolution approach to enhance efficiency and speed of outgrowth during single cell subcloning of Chinese Hamster Ovary cells. 2020 Computational and Structural Biotechnology Journal, 18, 1320–1329. | Google Scholar | CrossRef | PubMed
- Very high density of Chinese Hamster Ovary cells in perfusion by alternating tangential flow or tangential flow filtration in WAVE bioreactor™—Part II: Applications for antibody production and cryopreservation. 2013 Biotechnology Progress, 29(3): 767– 777. | Google Scholar
- Population Genetics The Stanford Encyclopedia of Philosophy [ Winter 2016 Edition], Edward N. Zalta [ed.], | Google Scholar
- Quasispecies theory in the context of population genetics. 2005 BMC Evolutionary Biology, 5, 44. | Google Scholar | CrossRef | PubMed
- Die Rolle von Serum bei der hydrodynamischen Belastung von tierischen Zellen im Bioreaktor—Möglichkeiten der Serum-Reduction, PhD thesis, ETH Zürich. | Google Scholar
- Diversity in host clone performance within a Chinese Hamster Ovary cell line. Progress, 31(5), 1186– 1200 | Google Scholar
- Single-cell transcriptome analyses reveal heterogeneity in suspension cultures and clonal markers of CHO-K1 cells. 2020 Biotechnology Bioengineering, 118, 944—951 | Google Scholar PubMed
- High level expression of tissue inhibitor of metalloproteinases in Chinese Hamster Ovary cells using glutamine synthetase gene amplification. 1990 Biotechnology, 8, 662– 667. | Google Scholar | CrossRef | PubMed
- Mammalian gene targeting with designed zinc finger nucleases. 2005 Molecular Therapy, 13(2), 438– 446. | Google Scholar | CrossRef | PubMed
- Sigma-Aldrich White paper: Accelerating cell line and process development | Google Scholar
- Rapid development of stable transgene CHO cell lines by CRISPR/Cas9-mediated site-specific integration into C12orf35. 2018 Applied Microbiology and Biotechnology, 102(14), 6105– 6117. | Google Scholar | CrossRef | PubMed
- Isoenzyme analysis as a rapid method for the examination of the species identity of cell cultures. In Vitro Cellular and Developmental Biology—Animal, 31, 115– 119. | Google Scholar | CrossRef | PubMed